What is the carbon footprint of bioethanol?
Being environmentally conscious is no longer a marginal concern. As the consequences of anthropomorphic climate change become more apparent, there is a growing collective desire to protect our planet and our environment. While we need people to change their behavior, both individually and at the societal level, to combat climate change, there is also a role for green technologies. Any innovation that allows us to reduce harmful emissions and generate less waste can be significant in the fight against climate change.
One of the main sources of global pollution is vehicle emissions. For many people, the use of a personal car is a necessity. Without a car, they wouldn't be able to go to work, take their children to school or travel much further than their own front door. But unless you can afford an electric car and are lucky enough to live somewhere with decent infrastructure, driving means contributing to the endless stream of pollution and greenhouse gas emissions we are all responsible for.
Bioethanol is emerging as a promising alternative fuel that could one day allow us to drive as much as we want without having to worry about what's coming out of our exhausts. Anyone striving for a carbon-neutral future should pay attention to bioethanol.
How is bioethanol fuel produced?
Bioethanol offers numerous advantages over conventional fuels, but its main selling point is how environmentally friendly it is. Bioethanol production uses renewable resources, namely crops such as corn, maize and corn. In principle, it can be produced from any plant that contains sugar and starch. We use the term "biomass" as an all-encompassing term for renewable organic materials derived from plants and animals. Biomass includes living organisms, materials derived from living organisms, and materials from organisms that have recently died.
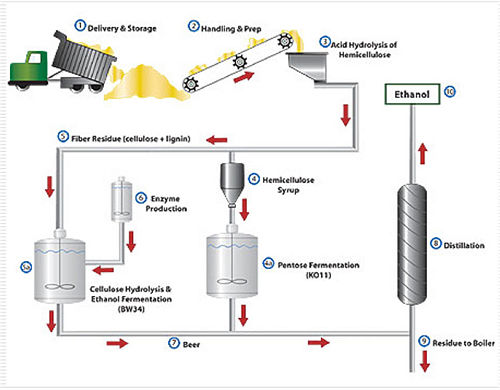
The exact process for producing bioethanol fuel depends on the composition of the biomass used. Most research is currently focused on lignocellulosic material, including willow, eucalyptus, straw, sugar cane, agricultural residues and other woody and grassy material often considered as waste. It takes 2 to 4 tons of lignocellulosic biomass to produce 1 ton of bioethanol. Researchers prefer lignocellulosic biomass because it is more abundant than food crops and cheaper to produce, especially since it is considered waste anyway. It also has a higher net energy balance than other forms of biomass, which makes it ecological makes it more attractive. Lignocellulosic biomass can reduce greenhouse gas emissions by up to 90%, which is much better than first generation biofuels.
The disadvantage of using lignocellulosic biomass for bioethanol production is that it contains a range of carbohydrate polymers, including cellulose. Cellulose is the main component of plant cell walls and is composed of glucose molecules. When cellulose is broken down in a process called hydrolysis, glucose is released. There are several ways to hydrolyze cellulose in lignocellulosic biomass to produce fermentable sugars. The most common methods include treating the biomass with acid or specific enzymes and heating it.
For non-lignocellulosic biomass, such as grain, the first step is to grind the biomass to release the starch. The resulting material is mixed with a specific volume of water to control the ratio of sugar to yeast in the resulting mash. Heating the mixture dissolves water-soluble starches, while acid or enzymatic hydrolysis is used to simultaneously convert the starch into sugars. The resulting mixture is bought up to a pH between 4.8 and 5.0, which is slightly acidic. This is necessary to allow the yeast that ferments the sugars to grow. The fermentation process produces ethanol and CO2. Subsequent distillation and dehydration increases the concentration of ethanol in the solution.
To be used as a fuel, ethanol must be very pure, as close to 100% as possible. Industrially produced ethanol has a relatively high water content that reduces purity. There are several methods of dehydrating ethanol, all of which are currently being investigated by researchers for efficiency improvements.
What are the environmental benefits of bioethanol?
Bioethanol not only comes from renewable sources, but also significantly reduces greenhouse gas emissions during production and when the finished fuel is burned, compared to conventional fuels. Adding ethanol to gasoline increases the octane number, which means it will burn rather than burn, and allows us to stretch our ever-shrinking oil reserves. Extending the life of oil reserves improves our fuel security and reduces our dependence on oil-producing nations.
If you're wondering if bioethanol produces carbon monoxide or other harmful substances, the answer is no. Burning bioethanol results in very few emissions; it is significantly cleaner than conventional fuels. The worst it gives off is a negligible amount of carbon dioxide, far too little to have any negative impact on human or environmental health.
You may have heard conflicting statements about the carbon footprint of bioethanol. So, is bioethanol carbon neutral or not? The answer is, yes, it is carbon neutral. But you have to ask yourself why bioethanol is carbon neutral. The answer is actually quite simple: the amount of carbon dioxide released in the production of bioethanol is the same as the amount of carbon dioxide taken up by the plants that produce the fuel during photosynthesis.
Switching from petroleum-based fuels to bioethanol will help heal the ozone layer over the earth, a long-term goal of the environmental movement. When ethanol burns, the resulting products are less reactive with sunlight than emissions from conventional fuel sources. Even large-scale burning of ethanol is unlikely to deplete the ozone layer.
Even if we continue to use conventional fuel for our vehicles in the near future, we can still use bioethanol to reduce greenhouse gas emissions. Blending bioethanol with gasoline offers a number of valuable benefits. First, it increases the octane number of the fuel, something that is usually difficult and expensive to do. By enriching low-quality fuel with ethanol, we can reduce harmful emissions and improve performance. Adding bioethanol to fuel also means we can further increase our supply and reduce our dependence on oil-producing countries.
Overall, widespread use of bioethanol would lead to improved air quality and a reduction in the prevalence of carcinogenic particles. Ongoing research and development projects are constantly improving the environmental benefits of bioethanol, making it an even more attractive prospect. For example, the latest designs of bioethanol plants ensure that all water discharged is environmentally neutral. Some plants recycle the waste water generated in the production of bioethanol so that it can be reused.
What is bioethanol fuel used for?
Ethanol is a versatile fuel that has already proven its potential in several key areas. The most prominent application of bioethanol today is as a transportation fuel, with many in the auto industry expecting it to eventually supplant gasoline as the primary transportation fuel for personal vehicles. However, one of the benefits of bioethanol is that it does not have to completely replace gasoline to make a difference. Ethanol is often used to enhance the performance of low-quality fuel, and there are several blended fuels on the market that combine bioethanol with diesel or gasoline.
Like any fuel, bioethanol can be burned to produce energy and generate power. Research into the potential role of bioethanol in electricity generation is ongoing. Compared to coal and oil, ethanol has a much lower thermal energy output, meaning that much larger quantities are needed to produce the same energy output. However, we can afford to burn much more ethanol without causing environmental damage and releasing toxic emissions.
Bioethanol fireplaces are also becoming increasingly popular. They don't produce smoke or other harmful byproducts, they look great, and they don't require a chimney or connection to a gas line.
How does bioethanol compare to conventional fuels?
Compared to gasoline, bioethanol has a much lower energy content. If you have two identical vehicles, one running on gasoline and the other on bioethanol, the car on gasoline will be able to continue running. However, bioethanol does not produce the same harmful emissions as gasoline. As the cost of producing bioethanol continues to fall and production methods become more efficient, we may well reach a point where having to fuel vehicles more often is considered a worthwhile trade-off.
Bioethanol has a higher octane number than gasoline and can even be added to gasoline to increase the octane number. A higher octane number means that bioethanol will burn rather than ignite in an engine and has superior anti-nuke properties.
Ethanol is less volatile than gasoline, as measured by Reid vapor pressure. The lower pressure of bioethanol means that it evaporates slowly, keeping the concentration of evaporative emissions low and further reducing the risk of the fuel exploding. However, the low vapor pressure can also be a disadvantage for ethanol. A combination of low pressure and a single boiling point means that engines running on pure ethanol cannot start unassisted at temperatures below 20 degrees Celsius. If bioethanol is ever seriously considered as a primary car fuel, engines will have to compensate for this problem.
Bioethanol and similar biofuels can play an important role in combating climate change. Not only does it have a wide range of potential applications, but the production of bioethanol is also carbon neutral. Keep an eye on this space, as it could well be the fuel of the future.